Report Published January 11, 2022 · 24 minute read
How to Improve Cancer Care and Lower Costs for Patients
Jacqueline Garry Lampert, David Kendall, & Ladan Ahmadi

Takeaways
Cancer rates and cancer deaths have been falling in the United States. And yet, cancer care is riddled with issues. The cost of care for patients is simply too high. Despite high costs, the quality of care varies widely with too much care that is plagued by racial and ethnic inequities and not evidence-based. In this report, we propose a new, innovative approach for Medicare to finance all cancer care through cancer care organizations (CCOs), which would be accountable for all aspects of cancer care, from the quality of care to patients’ costs. Medicare can drive changes across the health care system by encouraging private sector participation in CCOs.
“The medical establishment tells me I have ‘failed’ a number of therapies. That’s not right: The establishment and its therapies have failed me. The system we live in as metastatic breast cancer patients is simply not designed to deal with the cycle we are living and dying in,” wrote LA Times reporter Laurie Becklund shortly before she died of breast cancer.1 She spoke the truth about the frustration and disappointment of many cancer patients. All too often, cancer care fails patients after putting them through excruciating therapies. It is no one’s fault that cancer is very hard to prevent and cure, but so much of current treatment and financing is not leading to better outcomes and experiences for patients.
With cancer rates and deaths falling in part due to improved screening and smoking prevention, cancer care itself is the next big wave of improvement in the war on this disease. Current treatments and new ones on the horizon show great promise for curing cancer, but only if they are deployed effectively. And, right now, people of color are not experiencing the full extent of these gains, and costs are rising for patients and the whole health care system.
It is time to replace the fragmented payment system for cancer care with one that drives quality, equity, value, and innovation. In this report, we identify the key problems with cancer care in the United States and explain why current efforts are failing to fix care. We then propose a new, innovative approach that Medicare finance all cancer care through cancer care organizations (CCOs), which would be accountable for all aspects of cancer care from providers’ to patients’ costs, and encourage private health plans and employers to use them. This proposal will help achieve the goal of President Biden’s Administration to have all beneficiaries in traditional Medicare “in a care relationship with accountability for quality and total cost of care by 2030.2
Three Major Problems with Cancer Care
Cancer care in the United States has three major problems:
- The cost of cancer care for patients and the whole health care system is simply too high.
- Despite spending so much, the quality of cancer care varies widely.
- This quality variation leads to severe racial and ethnic inequities.
High Costs Overall and for Patients
The United States spends a lot of money on cancer care, and costs are rising. In 2015, the United States spent nearly $183 billion on cancer-related health care.3 These costs are expected to increase to $246 billion in 2030 based only on population growth, not considering potential high-priced advances in cancer treatment. It is no surprise that annual per capita health care expenditures for cancer patients are nearly four times higher than for patients without cancer.4 As cancer rates fall, overall spending for cancer care is growing more slowly than spending for all other disease categories. But the cost to treat each case of cancer is growing more rapidly than the average treatment cost for other diseases.5
The high cost of care is at least partially driven by prices for some cancer drugs that are too high given their value. For example, a value-based assessment of three chemotherapy drugs for recurrent ovarian cancer found prices were $6,900-8,850/month too high given their value, though some have questioned whether this analysis adequately included patient perspectives on the drug’s value.6
Cancer patients shoulder many of these costs themselves, out of their own pockets, regardless of insurance coverage status. Cancer patients paid $5.6 billion in out-of-pocket costs in 2018 alone, with average per patient costs of $3,600.7 However, per patient costs range widely, from $2,160-$31,200 per year.8 In many cases, the costs are prohibitive and a patient’s health suffers—a phenomenon known as financial toxicity, which is associated with worse outcomes and poorer quality of life.9 Health insurance is not necessarily protective against financial toxicity. A study of colon cancer patients, nearly all of whom were insured, found that about 25% of patients had treatment-related debt with an average debt of more than $26,000.10
While Medicare beneficiaries generally have better protection against costs, they do report financial toxicity, depending on the type of supplemental insurance in which they are enrolled. Medicare beneficiaries without supplemental insurance are fully exposed to a 20% coinsurance for most cancer care covered under Part B, including most chemotherapy, and a 5% coinsurance for Part D drugs when costs exceed the catastrophic coverage limit. Beneficiaries dually enrolled in Medicare and Medicaid experience the lowest average annual out-of-pocket costs ($2,116), while beneficiaries without any supplemental coverage experience the highest costs ($8,115).11
Costs are not borne equally by all cancer patients, and there are substantial racial, income, and educational disparities in the distribution of out-of-pocket costs. People of color and patients with lower incomes and less formal education were more likely to experience financial hardship.12 African American cancer survivors were more likely than white survivors to experience financial toxicity (31% versus 24%).13 Among breast cancer patients, Black women are more likely than white women (58% vs 39%) to report an adverse financial impact of cancer.14
Wide variation in cancer care
Cancer mortality has fallen 31% since 1991, mostly because Americans are smoking less and there are new and more effective screenings and treatments for certain cancers.15 For example, the death rate for lung cancer saw substantial improvement between 2014 and 2018, likely due to treatment breakthroughs for non-small cell lung cancer.16 Despite this, it is important to note that the death rate from some cancers like pancreatic, liver, mouth, and throat, is rising.
While a general decline in the death rate is good, cancer care still falls well short of where it should be. As UCLA oncologist and researcher Patricia A. Ganz has said, cancer care in the United States “falls short in terms of consistency in the delivery of care that is patient-centered, evidence-based, and coordinated.”17 We see this wide variation play out in different ways:
- Not all cancer care is accountable for outcomes that matter to patients. The way medical professionals and Medicare assess quality-of-care focuses on small details like the delivery of a specific service instead of the results of the care. A comprehensive set of oncology outcome-based quality measures has not been published, and outcome-based measures do not exist for many cancer specialties.18 The National Quality Forum’s Core Quality Measures Collaborative core measure set for medical oncology is focused mostly on process measures, and the Collaborative notes that patient reported outcomes and patient experience measures “remain a challenge and a priority area for oncology.”19
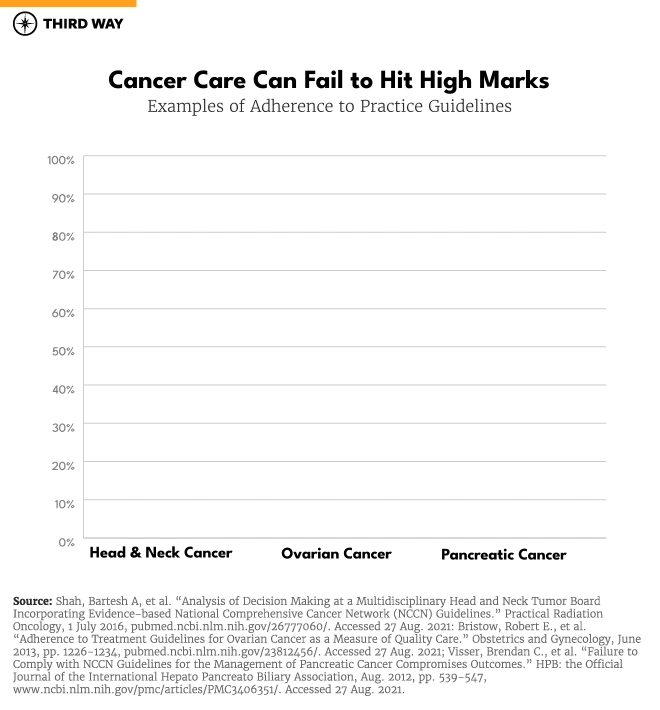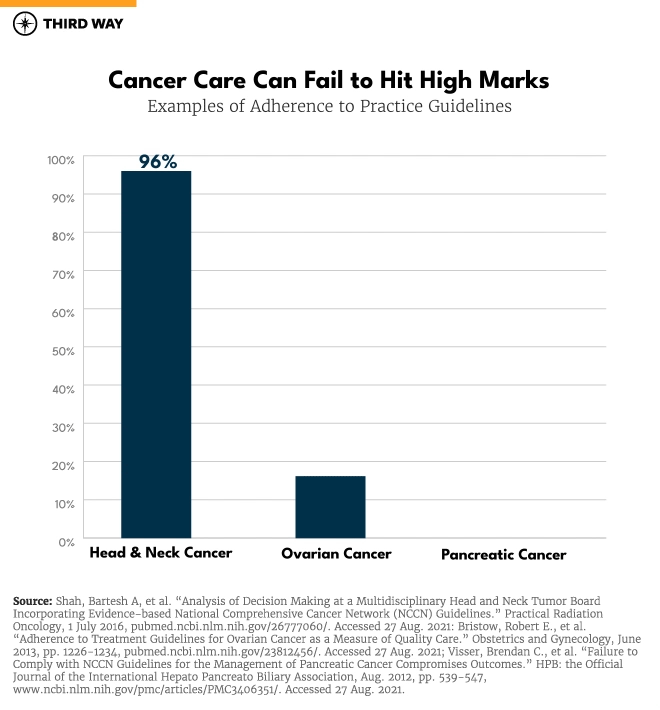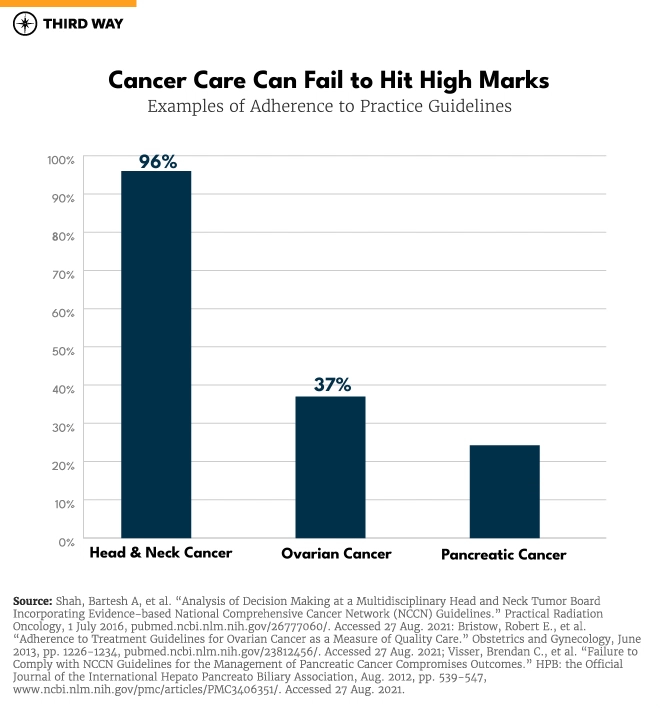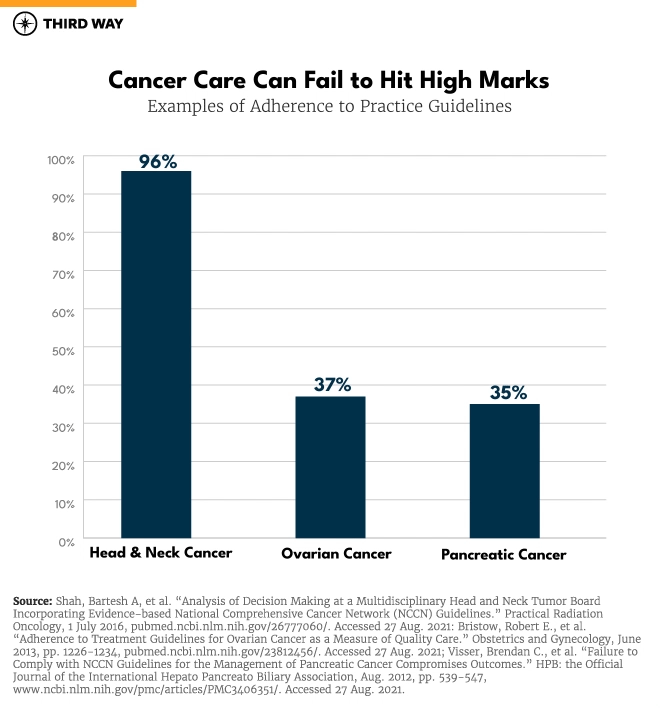
- Not all cancer care is evidence-based. Evidence-based Clinical Practice Guidelines exist for 97% of cancers in the United States.20 Unfortunately, adherence to the guidelines varies widely in oncology, ranging from as high as 96% for head and neck cancer21 to as low as 35% for pancreatic cancer22 and 37% for ovarian cancer.23 Adherence to the more than 70 Choosing Wisely recommendations for cancer care, which focus on tests and procedures that lack evidence and fail to add value, ranges from 53% to 78%, and the initiative has had little impact on reducing wasteful, low-value care delivered at hospitals and clinics throughout the country.24
- Not all cancer care is coordinated. 25 Cancer care remains hard for patients to navigate. Most cancer patients receive more than one type of treatment and must coordinate care received from a surgeon, oncologist, radiologist, and more. These providers may not be in the same clinic, practice, or even the same health system. The consequences of this lack of coordination can be catastrophic. As one study succinctly noted, “Poor care coordination during cancer treatment is associated with medical errors and sentinel events, poor symptom control, less-comprehensive supportive care, and increased utilization and costs.”26
Unequal Outcomes
This wide variability contributes to severe racial and ethnic inequities in cancer care and health outcomes.27 Racial and ethnic minorities, along with individuals of lower socioeconomic status and those who are uninsured, have a much higher incidence of certain cancers and are more likely to be diagnosed at later stages in their disease, which contributes to disparities in cancer survivorship.28 Racial and ethnic minorities are also less likely to receive standard treatment for cancer just as they are for health care in general.29 Here are some key examples:
- Black Americans are one-third more likely to die from cancer after accounting for other factors like age and stage of diagnosis.30
- American Indian and Alaska Natives are 51% more likely to die from cancer. 31
- Black men die from prostate cancer at a rate more than double that for all other men. 32
- Black women have a 40% higher breast cancer death rate than white women, despite having lower rates of diagnosis.33
- Hispanic Americans are less likely to die from cancer generally but have higher death rates for specific cancers like stomach, cervical, and liver.34
Research indicates the causes of these disparities are a complex combination of systemic and structural racism. Contributing factors include “barriers to high-quality cancer prevention, early detection, and treatment due to interrelated inequities in work, wealth, education, housing, and overall standard of living.”35 As an article for the American Society for Clinical Oncology succinctly puts it: “The differences in cancer survival are related to structural barriers to accessing care and overt documented differences in the delivery of evidence-based care.”36
Socioeconomic disparities in cancer are persistent and widening, particularly for preventable cancers.37 For example, death rates in the poorest counties in the United States are two times higher for cervical cancer and 40% higher for lung cancer (in men) and liver cancer than death rates in the wealthiest counties.38
Black men die from prostate cancer at a rate more than double that for all other men.
The COVID-19 pandemic will likely exacerbate these disparities as many Americans missed or delayed important cancer screenings due to public health measures to mitigate virus spread. For example, in April 2020, breast and cervical cancer screenings declined by 87% and 84% compared to five-year averages for that month, with even greater declines for breast cancer screening among American Indian and Alaska Native women (98%) and cervical cancer screening among Asian Pacific Islander women (92%).39 The consequences of these missed or delayed screenings may include cancer diagnosis at much later disease stages, a key factor in worse mortality rates.
Why Current Efforts are Falling Short
Current efforts have not yet taken a comprehensive approach to all aspects of cancer care—from coordination to quality—and overall costs for cancer patients.
For example, the Centers for Medicare & Medicaid Services (CMS) has not attempted a comprehensive cancer payment model that prioritizes and incentivizes coordination across multiple providers. Recent efforts by CMS have focused on patients receiving just one aspect of cancer treatment—just chemotherapy or just radiotherapy—when most cancer patients receive multi-pronged treatment. Specifically, the Oncology Care Model has focused on Medicare beneficiaries receiving chemotherapy.40
Despite not achieving overall savings through the first five performance periods, the model has been successful in important ways. Medicare Part A payments to providers under the model declined more than for those in a comparison group, and Part B payments to providers under that model grew more slowly than payments in a comparison group, both without hurting patient satisfaction.41 The payment differential has increased over time, suggesting that the model could eventually produce a net savings. In addition, the relative payment reduction has been greater in higher-risk episodes of care, where the Oncology Care Model has reduced Medicare Parts A and B payments below projections.42 Finally, this model has pioneered important new elements like a written treatment plan, which patients can study on their own time and not only when they are overwhelmed by information at a doctor’s office.
CMS has also finalized an alternative payment model using a bundled, episode-based payment for radiotherapy treatment.43 Radiation oncology accounts for just 3% of costs for actively treated cancer patients in Medicare, but fee-for-service payments to providers vary widely depending on the location where care is provided.44 The Radiation Oncology Model aims to address this while improving quality and adherence to evidence-based care. A major challenge is that CMS’ two cancer care Models don’t work in tandem, with the Oncology Care Model having to reconcile costs that are incurred under the Radiation Oncology Model. The lack of integration means there is no incentive for care coordination.
Another missing element in CMS’ payment models for cancer care is the price of drugs, which constitute a substantial share of the total cost of cancer care and are rising.45 Estimates of the portion of cancer costs dedicated to chemotherapy and other drugs range from 18-50%.46 Without addressing pharmaceutical prices, and the substantial share of those prices borne by patients, current models fail to address barriers that may jeopardize patient access to potentially life-saving medications.

The Solution
We need a new approach to cancer care—one which reduces costs and improves quality for all patients. To accomplish this, Congress should work with the administration to finance cancer care through cancer care organizations (CCOs), which would receive a value-based payment to improve the quality of care, lower overall costs and patient costs, and ensure equity among patients of different demographic backgrounds. Like an accountable care organization, the payment would cover health care costs while receiving treatment and follow-up care. To ensure continued community access to cancer care, CCOs would either be accountable for all health care costs or cancer costs only, depending on the experience and infrastructure of the CCO. Building on the CMS models, a CCO would give providers total responsibility for cancer care and would leverage Medicare’s market-leading power to drive change for all patients with cancer.
Creating CCOs will require the following five steps:
1. Develop outcome-based cancer care quality measures that matter to patients. Medicare should measure and pay for the kinds of things that matter to patients. For example, some patients may want to know how well they can expect to function during and after treatment, some may prioritize extending life as long as possible, while others may emphasize their quality of life and prioritize palliative or hospice care. To address this, the National Coalition for Cancer Survivorship facilitated a patient-driven measurement development effort focused on redefining functional status during treatment and into survivorship.47 Prioritized domains in this measure include global quality of life (including physical and mental health), physical function, pain, fatigue, cognitive function, and psychosocial illness impact (such as depression, independence, sense of control, and others).48 Quality measures should also reflect how effectively clinicians have engaged patients in shared decision-making about treatment options, including palliative, hospice, and end-of-life care.49 Ultimately, provider performance on these quality measures will, in part, determine payment rates under value-based arrangements described below.
To continue progress toward developing patient-centered, outcome-based cancer care quality measures, CMS should proceed with the quality measure lifecycle, using existing cancer care models and CCOs to collect data, refine measure specifications, define technical details, and more.50
Medicare should measure and pay for the kinds of things that matter to patients.
2. Use value-based payments to drive equity, quality, savings, and innovation. CCOs will have responsibility for the cost and quality of cancer care for assigned beneficiaries through value-based payment arrangements that drive equity and encourage innovative treatment options.
In order to maintain community access to cancer care, payments to CCOs will differ based on providers’ experience and infrastructure. Payments to larger CCOs with the infrastructure and experience to assume full responsibility for patient care will cover all Medicare items and services during a six-month episode of care. CCOs that meet quality and equity requirements will share in any savings they achieve. CCOs serving smaller communities will also be paid for a six-month episode of care, but payments to these providers will encompass cancer-related items and services only, with bonus payments for meeting quality and equity requirements.
The payment to CCOs for each patient will include pharmaceutical costs, which should incentivize CCOs to utilize lower-cost generics and biosimilars when available. Emerging data indicate biosimilar utilization is beginning to drive savings among the oncology care model participants.51 In addition, CCOs will be able to negotiate value-based payments for cancer drugs that are based on measurable benefits to patients.52 In the U.S., these arrangements are rapidly becoming more common between health plans and manufacturers, but medical groups are often not involved. In France, hospitals and hospital purchasing groups negotiate directly with pharmaceutical manufacturers to set the price for injected and infused drugs used in the hospital ambulatory or inpatient setting.53 A gainsharing framework with the French single-payer health care system encourages hospitals to negotiate the best possible price.
To begin progress toward value-based payment for prescription drugs, the Department of Health and Human Services (HHS) should collect data on off-label use of cancer treatments by CCOs to gather real world evidence on their performance and value. This could involve Congress working with states to require prescribers to note the specific indication or disease on their prescriptions or establishing confidential registries of off-label prescribing.54 In addition, Medicare should attribute Part B and Part D drug costs to patients and providers participating in the CCO. Finally, CCOs would be expected to advance the development and utilization of decision-support tools that integrate clinical and economic information into a single system for clinicians to use when discussing treatment options with patients.55
To ensure these changes improve care and outcomes for all patients, part of a CCO’s payment would include a reward or penalty based on health disparities. For example, if a CCO improved its quality scores for people of color as well for all its other patients, then it would receive a full bonus. As a first step toward tying payments to equity, CMS should require providers to submit relevant data, which will help advance work already underway to improve data collection and design interventions to advance health equity. A near-immediate use of such data could be to compare the racial and ethnic composition of a CCO’s patients to similar data of its community, to determine whether a CCO serves a patient population that reflects its community.

Finally, a fixed percentage of each CCO’s value-based payment will be earmarked for innovative preventive, diagnostic, and treatment methods, and for investments in infrastructure, such as data systems infrastructure that supports this new, comprehensive, coordinated model of care. For example, oncologists may wish to use new blood tests for detecting multiple cancers before they are officially approved for Medicare reimbursement.56 Medicare would base the amount for the innovation fund in part on economic analysis of the value of innovative methods and products as they become available for use. The decision about how to use the innovation fund would be up to the CCO. That way, the innovation fund would give clinicians flexibility to avoid conflicts between older treatments and experimental treatments as well as the opportunity to enroll patients in clinical trials concurrently.57
3. Remove statutory and regulatory barriers to value-based arrangements. Current policies aimed at preventing waste and fraud under the old payment system called fee-for-service are inhibiting the adoption of new, value-based payments. For example, the Anti-Kickback Statue and Stark Law prevent providers and suppliers from conspiring to share money gained from bilking Medicare but have also historically prevented suppliers from sharing technology that can enable providers to use their products more successfully. In addition, the Medicaid best price rule has had the unintended effect of limiting the scope of private sector arrangements that lower prices below what Medicaid currently pays.
A next step to clearing a regulatory path for value-based arrangements would be to fully apply recent regulatory exceptions to the Stark Law for value-based arrangements to the Anti-Kickback Statute, so that monetary remuneration is protected regardless of the amount of financial risk undertaken by participants. In addition, HHS should explore ways to permit pharmaceutical manufacturers to utilize recently created safe harbors from the Anti-Kickback Statute to help solidify legal certainty for entities seeking to participate in value-based arrangements, including CCOs. Such an effort should also support a new rule that ensures the value-based arrangements for drugs can move forward as allowed under the new Medicaid best price rule while ensuring state Medicaid programs have access to the same low prices through value-based arrangements and to non-value-based best prices for states that are unable to participate in value-based arrangements.58
4. Protect patients from high costs. Medicare would cap out-of-pocket costs for all patients based on income by filling gaps in existing programs like Medicare Part D for prescription drugs and the Medicare Savings programs for low-income beneficiaries.59 In addition, CCOs would have the flexibility to set patient cost-sharing in a way that optimizes access to care and recovery from cancer. Finally, CCOs would be permitted to use value-based insurance design to encourage use of high-value health care items and services and help reduce costs for patients with high health care needs. This would likely involve application of waivers for patient engagement incentives, such as the cost sharing support for Part B services available in some models tested by the Center for Medicare and Medicaid Innovation. Without such a waiver, the Anti-Kickback Statute would limit providers’ ability to waive copayments unless certain criteria are met.60
5. Sequentially phase-in the CCO model. To support participants’ success, we urge CMS to use an explicit plan with transparent criteria for moving through a series of steps like those outlined by the Health Care Transformation Task Force. Those steps include involving stakeholders in designing the model, prioritizing simplicity and transparency in model design, moving gradually to full financial responsibility for organizations managing the risk of paying for patients’ care, and carefully considering overlap with existing CMS payment models and ways to synchronize them.61
Conclusion
Cancer rates and cancer deaths have been falling in the United States, but cancer remains the second leading cause of death and the cost of treating cancer continues to rise.62 Cancer care is often high-priced and is not evidence based, meaning the nation does not get a good value for the cost. And variation in cancer care also produces unequal results for people of color and of lower socioeconomic status.
Recent efforts to improve cancer care in Medicare have helped in some cases but lack a comprehensive approach. Medicare should build on these efforts and create cancer care organizations (CCOs), which would be accountable for all aspects of cancer care, from the quality of care to patients’ costs. CCOs would receive value-based payments that reward providers for eliminating wasteful care, improving outcomes for patients equitably, and protecting patients from high costs. This CCO payment model would leverage Medicare’s leadership power to drive change for all patients with cancer.

Appendix - Barriers to a Comprehensive Approach to Cancer Care
In addition to piecemeal innovation models from CMS, several other issues impede a comprehensive approach to cancer care as noted below.
Existing outcome measures do not provide comprehensive accountability for the things that matter to patients. The Agency for Healthcare Research and Quality regards outcome-based measures as the gold standard in quality measurement. Yet process-based measures dominate many oncology quality assessment frameworks, including CMS’ Oncology Care Model and Radiation Oncology Model. Outcome measures that do exist do not focus on what matters most to patients.63 Measuring and reducing unnecessary utilization, such as hospitalizations and emergency department visits, is certainly important, but patients also care about how well they live with cancer and beyond.
Decision-support tools that integrate clinical guidelines and value-based recommendations when multiple treatment choices are emerging, thanks in part to demand created by the Oncology Care Model, but are not yet widely utilized. Value-based care benchmarks for cancer therapeutics have helped reduce costs but are not used across the entire health care system. While clinical guidelines help to define high-value, evidence-based care, they often include multiple treatment choices with no preferential order.64 The challenge remains to move from practice guidelines to a clear value metric for a specific cancer therapeutic. In the U.S., the American Society of Clinical Oncology (ASCO) Value Framework and National Comprehensive Cancer Network (NCCN) Evidence Blocks attempt such a definition, aiming to help clinicians and patients choose between existing, comparable treatments, but both have shortcomings.65 The Institute for Clinical and Economic Review (ICER) framework considers clinical and economic data to measure how well a particular intervention improves and lengthens patients’ lives. ICER takes into account both long-term value and short-term affordability to set a value-based price benchmark.66 Using this framework, ICER has found that chimeric antigen receptor t-cell (CAR-T) therapies are cost-effective for certain patients despite wholesale acquisition costs between $375,000-475,000.67 On the other hand, ICER found that discounts of 57-78% off the costs of several ovarian cancer maintenance drugs were required in order to meet cost-effective benchmarks, despite those drugs having a much lower cost than the CAR-T therapy.68 The ideal decision-support tool would seamlessly integrate clinical and value decisions into a clinician’s workflow and enable them to engage patients in decision-making about treatment choices.69
Routine evidence development and data collection of off-label uses of oncology drugs are largely nonexistent. Researchers estimate that more than half of oncology drug use is off-label.70 Off-label use of a drug means, for example, it is prescribed by a clinician for an indication, at a dose, for a duration, or for a patient different from the precise requirements for which the Food and Drug Administration (FDA) approved the drug. Because the FDA regulates the drug and the manufacturer, but states regulate clinicians and pharmacies, physicians may generally (except in certain restricted circumstances) prescribe an FDA-approved drug for uses beyond the label.71 Medicare and many commercial insurance payers cover drugs used off-label so long as the off-label uses are included in an approved drug compendium.72 However, states do not require physicians to include on a prescription the reason for prescribing a drug, which makes it challenging for existing databases to determine when a drug is being used off-label. One helpful development is a recent draft FDA guidance on ways to standardize the use of data from the off-label use of drugs and other uses, which could lead to more evidence development and data collection.73
Efforts at paying for value are limited by statutory and regulatory barriers. Several significant statutes and regulations make implementation of value-based health care payment models and contracts challenging for a wide variety of health care organizations.74 In late 2020, the Trump administration finalized regulatory changes to the Stark Law and Medicaid best-price rule to provide greater flexibility and help advance greater adoption of value-based payment arrangements.75 However, significant hurdles to value-based care arrangements between payers and providers remain. The Anti-Kickback Statute inhibits the creation of value-based payment and contracts in all segments of the health care industry. In addition, FDA rules and policies on communication by pharmaceutical and medical device manufacturers about medical products limit, in certain circumstances, the assessment of clinical benefits of care, the sharing of cost information, and the degree of cost savings.
Patients do not have comprehensive protection from financial toxicity. Health care costs present a serious anxiety for most Americans, even those who have health insurance coverage.76 Current efforts, including proposed limits on drug prices and the recently enacted ban on surprise bills, are good and important, but do not comprehensively address a patient’s total cost problem.77 They do nothing to guarantee that patients’ costs will not continue to explode. And they do not control all the other underlying causes of high costs including wasteful care, high provider prices, and administrative overhead.78
Data on racial and ethnic disparities is incomplete and existing data highlight a health care system that is failing people of color. Through Executive Orders, President Biden has directed federal agencies to begin work on foundational data challenges and Congress is also working on standards and definitions.79 This work is in early stages and must be continued to accurately measure disparities and design interventions to eliminate them.


